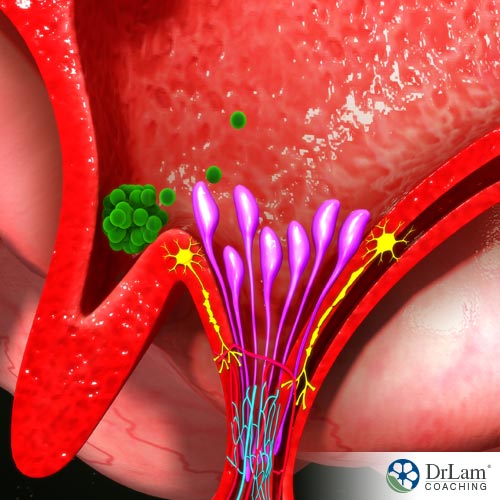
 Everyone interested in Adrenal Fatigue Syndrome (AFS) is aware of the importance of the hypothalamic-pituitary-adrenal (HPA) hormonal axis in the body’s natural response to stress. This HPA axis sets in motion the cascade of responses that results in the adrenal glands releasing cortisol to fight the effects of stress on the body. In the brain, hypothalamus hormones start the sequence of other hormones being brought into play in the stress response.
Everyone interested in Adrenal Fatigue Syndrome (AFS) is aware of the importance of the hypothalamic-pituitary-adrenal (HPA) hormonal axis in the body’s natural response to stress. This HPA axis sets in motion the cascade of responses that results in the adrenal glands releasing cortisol to fight the effects of stress on the body. In the brain, hypothalamus hormones start the sequence of other hormones being brought into play in the stress response.
Ultimately, if stress is unresolved, adrenal fatigue can result. Once this fatigue sets in, not enough cortisol is released to deal with the effects of stress. Thus begins the set of symptoms frequently seen in AFS.
These symptoms are vague at first and not often seen as being connected by traditionally trained physicians during their evaluation of patients presenting with them. Symptoms such as continuing fatigue even after sleeping well all night, gaining weight around the middle of the body, not able to go to sleep, feeling anxious and depressed, suffering from allergies easily, difficulty concentrating, and brain fog.
Biochemically, as AFS progresses, cortisol levels tend to first rise but eventually fall. Your body responds to the lower level of cortisol in several ways. Stress will lead to your gut system developing what is called leaky gut, in which toxins, bacteria, and viruses leak from the gut into your bloodstream and throughout your body. This leads to increased inflammation and an overactive immune response.
In women, estrogen dominance begins playing a role in health, as cortisol precursors (such as progesterone) may be shunted towards cortisol synthesis. The reduction of progesterone can lead to a relative increase in estrogen, or a state called estrogen dominance. Symptoms include PCOS, endometriosis, fibrocystic breasts?, fibroids?, and even estrogen driven cancers.This overabundance of estrogen sets them up for very significant chronic illnesses.
Estrogen also may play a vital role in protecting women from some of these very chronic illnesses that can be brought on by an excess of the same hormone.
The Women’s Health Initiative (WHI) was an ambitious study designed to investigate effects of synthetic hormone replacement therapy with estrogen on women’s health. Its negative findings were significant enough to stop the study early. A major finding of this study was an increased risk of several serious medical conditions including heart attack, stroke, and breast cancer in women taking a specific combination of hormones.
These findings encouraged women to stop synthetic hormone replacement therapy and encouraged physicians to consider whether they would recommend this therapy to their patients. Unfortunately, the recommendations were not specific to the estrogens used in the research. Perhaps, this may have resulted in some women coming off needed hormone replacement therapy when they may not have needed to do so.
Several shortcomings in this study suggest the findings may not be as generalizable as they have been considered.
One significant shortcoming had to do with the choice of hormones investigated. Premarin is a conjugated equine estrogen made up of hormones and hormone metabolites from pregnant mare urine. Prempro is an estrogen-containing medroxyprogesterone acetate, a substance known to cause birth defects. Both of these are synthetic hormones and are not natural to the human body.
 Estradiol is the preferred naturally occurring estrogen for replacement therapy. Using a synthetic hormone or hormones from other species such as Premarin won’t give the desired results in a human female as compared? to natural estrogens such as estradiol or estriol.
Estradiol is the preferred naturally occurring estrogen for replacement therapy. Using a synthetic hormone or hormones from other species such as Premarin won’t give the desired results in a human female as compared? to natural estrogens such as estradiol or estriol.
Another shortcoming in the WHI research was the dosage amount utilized. Low doses on a continuing basis with no consideration of the physical needs of the women regarding estrogen simply were not sufficient for the desired purpose. Estrogen fills many roles in the female body and levels vary, sometimes considerably, during a woman’s monthly cycle. Disregarding this changing need at different times in women’s physiology, even women going through or post-menopause, was a confounding factor in this research.
Yet another factor that limited the validity of results in this research was the delivery system.
This may have been the most serious limitation of the study because of some of the potential results of taking estrogen by mouth. These potential results include an increase in clotting factors that may bring about increased risk of blood clots, DVTs, pulmonary embolism, and stroke.
There could also be an increase in hormone binding proteins, such as SHBG, that could lead to significant detrimental physical results. An increase in mediators of inflammation, such as pro-inflammatory cytokines, could also result from orally administered estrogen. Increased blood pressure and weight gain are possible as well. One very significant, even critical, problem with orally administered estrogen therapy is the inhibition and reduction of human growth hormone.
These very significant problems with the WHI research may have led to the findings that prompted so many women to stop synthetic hormone replacement therapy on their own or to go to their physicians and request a stop in replacement therapy. Current research suggests strongly that estrogen is a necessary hormone for a number of reasons and that women whose estrogen levels are low due to menopause or other reasons may do well with replacement therapy – if administered correctly.
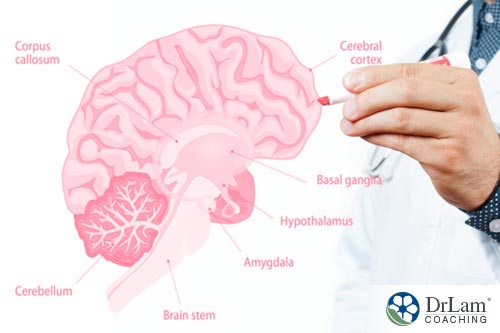
Hypothalamus hormones play several roles in women’s health. In general, hypothalamus hormones affect metabolism and the autonomic nervous system. While these hormones regulate body systems that operate within all people, there are specific connections with women’s reproductive systems and estrogen and these hormones controlled in part by the hypothalamus.
Estrogen and progesterone have an effect on gene expression and on activating protein synthesis through the estrogen and progesterone receptors in neurons in the anterior and mediobasal areas of the hypothalamus, respectively. These estrogen-sensitive neurons in the hypothalamus respond differently in males and females.
Estrogen also plays a major signaling role in hypothalamus hormones. It affects signaling in the major neuroendocrine and autonomic cellular groups of the hypothalamus. In this way, it plays a part in the releasing of the hypothalamus hormones as well.
Hypothalamus hormones also affect reproduction in females. Estrogen and luteinizing hormones operate in a positive feedback loop to prepare the uterus for implantation of a fertilized egg. Once an egg is released, progesterone inhibits the hypothalamus and anterior pituitary to stop this feedback loop.
If there is no conception, levels of progesterone side effects decrease, stimulating the hypothalamus to secrete GnRH, gonadotrophin releasing hormone, to start the woman’s cycle over again.
This relationship between the hypothalamus hormones and estrogen is important for another reason, also. Recent research has shown estrogen to have a neuroprotective function in regard to protecting the brain and its neurons from inflammation and in the repair and remodeling of the brain. Appropriate levels of the correct types of estrogen will assist in the function of the hypothalamus through its protection of neurons.
 With the rapidly increasing incidence of serious chronic health conditions – such as Alzheimer’s Disease, Parkinson’s Disease, and stroke – researchers and clinicians are eagerly seeking ways to prevent or lessen the severity of these conditions. The increasing length of human life spans makes these kinds of conditions more likely in the future.
With the rapidly increasing incidence of serious chronic health conditions – such as Alzheimer’s Disease, Parkinson’s Disease, and stroke – researchers and clinicians are eagerly seeking ways to prevent or lessen the severity of these conditions. The increasing length of human life spans makes these kinds of conditions more likely in the future.
Alzheimer’s affects roughly 4 million people in the U.S. Nearly half of those over 85 suffer from this condition. It is the most common cause of dementia in the U.S. for people over age 65. The number of people with this condition doubles for every five years of age over 65.
Parkinson’s affects about 1 million people in the U.S. One in 20 people over the age of 80 have Parkinson’s Disease.
These numbers indicate the severity of just two of the serious chronic health conditions that can be helped by estrogen of the right kind and in the right dose at the right time. Significant research has shown this hormone to help protect women from these aging conditions.
Animal research has shown estrogen to reduce the amount of neuronal death in rodents with stroke-like symptoms, traumatic brain injury, and Parkinson’s-like symptoms. Estrogens thus appear to have direct effects on neurons, although the exact mechanisms are just now being investigated and defined.
Some of these mechanisms may involve activated nuclear estrogen receptors, some of which are located in the hypothalamus, altering the expression of some proteins, activating the mitogen-activated pathway, and direct antioxidant activity.
Estradiol, the primary estrogen in humans, has been shown to delay onset or decrease the risk of Alzheimer’s and other neurological illness conditions. It also lessens the amount of cell death in conditions like stroke or brain injury.
Two mechanisms appear to be at work when estradiol lowers the amount of injury in these brain conditions. It may directly attack the neurotoxic stimulus that results in the illness conditions, or it may increase the ability of the brain to regenerate and repair itself after the injury or illness condition.
One factor that increases the importance of estrogen’s neuroprotective function is the increasing age of women in the U.S. As more women age, their risk of stroke and other neurological disorders increases. One reason for this increase is that the age of menopause remains roughly the same.
Therefore, more postmenopausal women are living longer, meaning without the protective effects of naturally-occurring estrogen on their brain functions. They also will have more years without estrogen’s protection of the neurons of the hypothalamus. This will lower the amount of hypothalamus hormones available for these women.
 Prior to the time of menopause, women appear to be more protected from the risk of cerebrovascular accidents than men, likely due to the influence of a higher level of estrogen and other hormones controlled by the hypothalamus in their bodies. During and following menopause – as the levels of estrogen decrease – the incidence of stroke in women increases.
Prior to the time of menopause, women appear to be more protected from the risk of cerebrovascular accidents than men, likely due to the influence of a higher level of estrogen and other hormones controlled by the hypothalamus in their bodies. During and following menopause – as the levels of estrogen decrease – the incidence of stroke in women increases.
Clinical studies have shown the outcomes of these strokes in postmenopausal women to be worse than those of men. Disability and fatality rates are significantly higher for these women. Thus it appears the lower levels of estrogen and possibly other hypothalamus hormones decrease the protective factor enjoyed by women of pre-menopausal age.
In support of this contention, animal studies have shown estrogen to dramatically decrease the severity of stroke damage. This result does not appear to be due to any effect of estrogen on blood flow in the brain, but rather to its effect directly on the brain.
One of the hypothalamus hormones, the ovarian hormone 17beta estradiol (E2), performs a crucial role in many physiological functions. It operates through the estrogen receptors found throughout the brain. The most active form of estrogen, E2 may even work independently of the estrogen receptors in the brain. This would make the time for its effects much shorter, thus decreasing the effects of stroke faster.
Among the many effects of E2 on the brain are significant influences on memory formation, cognition, and motor coordination. All of these are detrimentally affected by the loss of function due to stroke. The application of E2 would appear to decrease the loss of function in these areas through its neurocognitive effects and the increased ability of the brain to repair and remodel itself following a stroke.
 Demonstratively the main cause of age-related dementia, Alzheimer’s is the reason behind at least 50 percent of dementia cases. It is a complex of declining hypothalamus hormones, cognitive and functional skills beginning in middle age or later and includes behaviors that vary in severity and type.
Demonstratively the main cause of age-related dementia, Alzheimer’s is the reason behind at least 50 percent of dementia cases. It is a complex of declining hypothalamus hormones, cognitive and functional skills beginning in middle age or later and includes behaviors that vary in severity and type.
Both dementia and a clear pattern of changes in the brain are present. Some of these brain changes include atrophy, loss of neurons, extracellular plaques that contain beta-amyloid peptides, intracellular neurofibrillary tangles, and granulovacuolar cytoplasmic changes in several brain regions.
Estrogen plays a significant role in some of these brain regions affected by Alzheimer’s. This suggests the lower levels of estrogen in aging brains in both men and women could play a part in the development of this health condition.
Projections by scientists involved in studying Alzheimer’s are that the number of people who suffer from this condition will increase to 14 million in the U.S. by the middle of this century. A connection has been shown between the effects of menopause in women and Alzheimer’s. The onset of menopause signals a decrease in protection of the brain by hypothalamus hormones and estrogen. Currently, three times more women than men have Alzheimer’s.
Interestingly, research into the relationship of Alzheimer’s and estrogen has shown mixed results. Reasons for this are unclear but may involve different forms of estrogen, different dosages, and different times and ways of administration.
One of the factors that must be taken into consideration when evaluating the effects of estrogen or any other hypothalamus hormones is the bioavailability of the hormone. Bioavailability levels may be much different than total levels of any hypothalamus hormones or estrogen. A hormone that is present but not usable by the body will not have the beneficial effect desired.
A large part of the estrogen circulating in the body is bound to sex hormone binding globulin (SHBG), which is one of the hypothalamus hormones. This estrogen does not cross the blood-brain barrier and thus doesn’t have the beneficial neuroprotective effect of the non-SHBG bound estrogen.
There has been research indicating increased levels of SHBG to be associated with the cognitive decline seen in Alzheimer’s. This connection supports the findings related to estrogen having a positive effect on Alzheimer’s symptoms. Some studies suggest low bioavailable estrogen and high SHBG post-menopause may increase the likelihood of Alzheimer’s developing.
However, other research has either not found similar effects of estrogen on cognitive outcome or found the opposite. Reasons for these discrepancies may involve the different procedures used in measuring hormones or the difficulty in measuring estradiol in the bloodstream of women who have been postmenopausal for many years.
Another possible reason for these apparently conflicting results may have to do with the type of estrogen utilized in the studies. A study conducted at Stanford University examined two groups of women – one of which took estrogen replacement therapy and the other stopped it.
These women, ages 50 to 65, had metabolic activity in specific brain regions involved in memory and executive function studied using PET scans. These are areas of the brain that can show deterioration before signs of Alzheimer’s appear.
The women were also chosen because of their increased risk of developing Alzheimer’s due to having a first-degree relative with the condition or due to having the specific variant of the ApoE gene that has been shown to be associated with Alzheimer’s.
 Results of this study showed that women who were taking estradiol as their replacement hormone had better preserved brain function than women taking Premarin, a conjugated equine estrogen. Women who were taking Premarin saw a decline in functioning in the brain areas investigated.
Results of this study showed that women who were taking estradiol as their replacement hormone had better preserved brain function than women taking Premarin, a conjugated equine estrogen. Women who were taking Premarin saw a decline in functioning in the brain areas investigated.
Along with estrogen, some of the women in the study also took progesterone. This is fairly common with hormone replacement therapy due to the increased risk of uterine cancer in women who only take estrogen. In this study, the progesterone cream appeared to significantly decrease the benefits seen with estradiol and increased the deficits seen with Premarin.
There appear to be benefits of taking estradiol as a hormone replacement, but not with progesterone. The benefits were much more significant than any seen with the conjugated equine urine estrogen. Much more study is needed before these benefits can be deemed certain.
Some possible shortcomings of this study include lack of randomization and the use of metabolic changes in some brain areas. The first suggests these same findings might not be seen in a larger, better-controlled study. The second suggests changes in those brain areas are only possible precursors of Alzheimer’s and not a sure sign of the condition.
Another possibly important factor to be considered by women choosing hormone replacement therapy is whether to take progesterone with their estrogen replacement. The findings of this small study are very interesting and, if proven accurate through more research, would suggest the risk of developing Alzheimer’s might be increased with the addition of progesterone to hormone replacement therapy.
Other research has investigated the effect of insulin-degrading enzyme (IDE). This protein degrades beta amyloid, another protein fragment associated with symptoms of Alzheimer’s. A decreased level of estradiol, such as happens during menopause, decreases the effectiveness of IDE. This would lead to more beta amyloid because the brain can’t rid itself of this protein.
Researchers also found the timing of the use of estrogen to be important in its effects. In general, the earlier estradiol is added, the more beneficial its protective function. Beginning estrogen therapy at the beginning of menopause increases the benefits to the woman’s brain and decreases her risk of developing Alzheimer’s later in life. Women who started estrogen replacement in their 60s did not see the benefit seen by women who started it in their 40s or 50s.
In addition to timing, the dosage of the hormones used is important to producing hypothalamus hormones. Using a naturally occurring estradiol brings a better response than using synthetic estrogen. The synthetics are not metabolized properly due to their chemical makeup and thus can be seen as toxins by the woman’s system.
Bioidentical estrogen absorbed through the skin appears to bring better results than a synthetic estrogen taken orally. The same appears to be true of bioidentical progesterone. This type of progesterone serves to protect brain functioning where synthetic progesterone may hamper functioning.
Current research has shown men also have a hypothalamic-related hormone that serves a protective function. Testosterone has been studied in applications with players in the National Football League who suffered concussions as well as with wounded warriors from Iraq and Afghanistan.
Concussions typically lead to hypopituitarism, in which the pituitary gland fails to produce sufficient hormones. This leads to a disruption in the HPA axis, a significant part of the body’s response to stressors. Insufficient levels of testosterone can result, leading to deficiencies in cognitive function and hypothalamus hormones.
 Studies have shown those who suffer from concussions or other traumatic brain injuries recover cognitive functioning faster and to a greater extent with higher testosterone levels. Bioidentical testosterone replacement appears to be the best choice for this type of remediation.
Studies have shown those who suffer from concussions or other traumatic brain injuries recover cognitive functioning faster and to a greater extent with higher testosterone levels. Bioidentical testosterone replacement appears to be the best choice for this type of remediation.
Increasingly, low levels of testosterone in men have been linked closely with the development of Alzheimer’s. In fact, some research has indicated men have a three times greater risk of Alzheimer’s if their testosterone levels are low.
Testosterone appears to provide the same neuroprotective functions as estrogen. Evidence from research indicates testosterone to improve the axonal regeneration and survival of motor neurons. It has been linked to an increase in neuronal growth, plasticity, and in the development of new synapses in the spine and pelvic regions.
Research into whether androgens like testosterone provide a neuroprotective function have seen interesting results.
One study of the effects of androgens on dopamine neurons under oxidative stress showed pretreating these cells reduced oxidative stress-induced cell death, but using androgens after the oxidative stress actually increased cell death. This suggests using androgens when oxidative stress levels are low is neuroprotective, but the opposite is true under conditions of high oxidative stress.
Because the male brain is very dependent on testosterone to function properly, low levels of these hypothalamus hormones can result in cognitive decline, lessened memory ability, and other dementia-related symptoms. Low testosterone levels have also been associated with increased secretion of beta-amyloid proteins which have been implicated in the development of Alzheimer’s.
Acetylcholine, a neurotransmitter dealing with learning and memory, is dependent on testosterone as well. Low testosterone would lead to lower levels of this neurotransmitter and difficulty with learning and memory. Dopamine, a neurotransmitter that appears to protect the brain against Parkinson’s Disease, is also dependent on testosterone. Low testosterone has been implicated in the development of Parkinson’s.
There are some potential drawbacks of testosterone therapy, also. This hormone has been shown not to have neuroprotective functions in either the methamphetamine-induced neurotoxicity of the dopaminergic system in rats nor in glutamate-induced neurotoxicity.
The use of testosterone cream may also lead to problems. This use must be closely followed to assure the dosage is not too high. Too high levels of testosterone in this form can be stored in body fat. This causes a disruption between hormone glands and the brain and also can lead to resistance at the hormone receptor site. A metabolic imbalance will occur, leading to symptoms of low testosterone because the hormone won’t get absorbed into the cells.
For some men, low testosterone is caused by insulin surges over a long period of time due to ingestion of large amounts of sugar and carbohydrates. This causes stress on the body, lowering testosterone and increasing the risk of dementia and Alzheimer’s. By balancing blood sugar and reducing inflammation, some men may get a return of natural testosterone from their body.
In some cases with older men who already have symptoms of one of the chronic neurodegenerative illness conditions, testosterone replacement therapy may help improve quality of life. For others, however, the testosterone is converted into estrogen through a process called aromatization. This is often seen when there is high blood sugar, insulin resistance, and prediabetes present. A brain-healthy diet can improve this situation and prevent aromatization.
It would appear the first step in changing testosterone levels is to make dietary changes to see whether the body will return to making testosterone itself. If this fails, bioidentical testosterone should be the one to use.
 Estradiol has been shown to be effective in preventing the degeneration seen in women at risk for developing dementia. This remediation effort appears to be most effective when started soon after menopause.
Estradiol has been shown to be effective in preventing the degeneration seen in women at risk for developing dementia. This remediation effort appears to be most effective when started soon after menopause.
Investigators at the Stanford University School of Medicine also found the use of Premarin, another synthetic estrogen replacement, not to have this protective factor. There are over 30 substances in Premarin, which is produced from pregnant mare urine. Only 17 percent of Premarin is estradiol. This study looked at brain metabolism as it is affected by stopping or continuing hormone therapy.
Findings showed the neurological effects of hormone therapy on women at risk of dementia depended critically on when the hormone therapy was started and on whether estradiol or Premarin was used.
Metabolic activity in the area of the brain in and around the hippocampus was preserved when women were started on estradiol within a year of menopause and stayed on it. However, when this regimen was stopped, the metabolic activity decreased significantly.
Women who were started on Premarin at the same time experienced more metabolic decline. When progestin, a synthetic progesterone, was added to either estradiol or Premarin, the decline was worse. With estradiol, the progestin countered totally the neurological benefit of estradiol. With Premarin, the metabolic decline increased dramatically.
It does appear that estradiol started when the beginning signs of dementia are seen has a neuroprotective function in women. This function is so robust, it could put off dementia if started early enough.
Another study at Stanford University supported these findings. A number of women under 60, in good overall health, who started hormone replacement therapy within a year of onset of menopause were studied. All were at increased risk of development of dementia due to having either a history of major depression, a first-degree relative with Alzheimer’s, or are positive for the ApoE4 allele. This gene variant is well known for increasing women’s risk of Alzheimer’s.
 PET scans at the beginning of the study and again two years later were obtained. Those women who continued on the estradiol therapy showed metabolic activity in the medial prefrontal cortex to be better preserved than those women who stopped the therapy.
PET scans at the beginning of the study and again two years later were obtained. Those women who continued on the estradiol therapy showed metabolic activity in the medial prefrontal cortex to be better preserved than those women who stopped the therapy.
Metabolic activity in another brain area, precuneus/posterior cingulate region, important in predicting visible signs of dementia, was very well-preserved in women who remained on the estradiol regimen. However, in women who stopped the therapy, this region was very negatively affected.
Women in Premarin usage showed no lessening of deterioration of metabolic activity in this region. If progesterone was added to either replacement hormone, the results were significantly worse.
While estradiol has very beneficial effects on brain preservation, there is also an increased risk of breast and uterine cancer. Women should consult their primary health care professional to determine risk versus benefit.
 Estrogen dominance and AFS are closely related. Late stage AFS often is associated with estrogen dominance in the symptom presentation. In the earlier stages of AFS, as the adrenals are under considerable burden to output cortisol to fight the effects of stress, more and more progesterone is needed. Progesterone is a precursor to cortisol. The more cortisol is synthesized, the more progesterone is needed. Natural hypothalamus hormones work best for the body.
Estrogen dominance and AFS are closely related. Late stage AFS often is associated with estrogen dominance in the symptom presentation. In the earlier stages of AFS, as the adrenals are under considerable burden to output cortisol to fight the effects of stress, more and more progesterone is needed. Progesterone is a precursor to cortisol. The more cortisol is synthesized, the more progesterone is needed. Natural hypothalamus hormones work best for the body.
Since estrogen and progesterone work as a team, balancing each other, the situation described above sets the stage for relative estrogen levels to increase leading to estrogen dominance.
Lowering estrogen levels is a prime consideration in remediating AFS. Working to heal the adrenals will make more progesterone available, thus balancing estrogen. One way to heal the adrenals is to lower overall stress, thus relieving the burden on the adrenals.
Balancing estrogen with progesterone is best achieved with natural progesterone, provided that the body can tolerate it. The synthetic progestin is not a good substitute for natural progesterone for the reasons enumerated above.
Type of administration of hormones is important in remediating imbalances found in AFS. Orally administered synthetic estrogen is not the preferred pathway for this hormone. Reasons for this have been discussed above. Most women find the low-dose topical pathway the best. It’s both inexpensive and effective. Especially in cases of AFS, the right administration method is important.
Dosage is likewise important. The wrong dose or wrong timing of doses can increase the symptoms of AFS and possibly even bring about adrenal crashes. Even normal doses may be excessive for the person with AFS. Adrenal crashes can be triggered. The weaker the body, the greater the risk.
Those with systemic congestion at the liver or extracellular matrix are particularly prone to negative outcome. Hormone replacement, even natural forms, is not for everyone. Due to the severity of symptoms of estrogen dominance, it is tempting to rush administration of hormones. It’s much more efficient and effective to heal the adrenals first, then administer hormones. This gives the liver more time to heal as well so it can metabolize the hormones given.
 The best way to administer natural progesterone is subject to some disagreement. Most women take the hormone by mouth because this comes as a prescription from their healthcare professional. The liver metabolizes about 80 percent of the hormone, so a significant dose must be taken in order to get a beneficial dose. These large doses will put extra stress on the liver.
The best way to administer natural progesterone is subject to some disagreement. Most women take the hormone by mouth because this comes as a prescription from their healthcare professional. The liver metabolizes about 80 percent of the hormone, so a significant dose must be taken in order to get a beneficial dose. These large doses will put extra stress on the liver.
People with AFS already have a low clearance state for progesterone with high reactive metabolite load. An overload of progesterone through the oral route will overwhelm the liver and possibly bring on an adrenal crash as mentioned before.
Because of this, the oral route of administration of progesterone side effects is not recommended for those with AFS unless properly supervised.
Testosterone is another of the hypothalamus hormones that become unbalanced with AFS. Lower levels of testosterone are related to decreased libido and sex drive in both men and women. This is also common in AFS. Replacement of testosterone can help. However, the administration of testosterone should not be too aggressive, because it can lead to overstimulation and adrenal crashes.
When menopause occurs, the symptoms sometimes become difficult to deal with. Women seek help to alleviate the symptoms, but in the recent past have been counseled not to use hormone replacement therapy.
Research has indicated that hormone replacement therapy is effective, but some hormone replacements have not proven effective. Bioidentical hormones have been shown to be effective due to their bioavailability, but the benefit is not universally evident. Hormone replacement of any kind needs to be personalized Careful measurement of hormone levels and continued monitoring are necessary.
Both men and women need testosterone to function optimally. Libido, endurance, and mood are all affected by testosterone levels. With AFS, the appropriate level of testosterone can alleviate some of the discomforts felt by both men and women. Excessive testosterone can trigger adrenal crashes due to the anabolic and stimulatory nature of this compound.
Progesterone is another of the hypothalamus hormones necessary for women to achieve relief from symptoms of both menopause and AFS if there is a state of estrogen dominance.
In cases of AFS, progesterone is often depleted in the production of cortisol. This leads to estrogen dominance and all of the symptoms of that condition. Progesterone is needed to balance estrogen in both menopause and AFS. Once again, dosage and type of administration are important.
A deficit of estrogen can be seen in the later stages of AFS as well, especially in those who are in catabolic state.
Supplementation will help women with their symptoms if this is the case. Care must be taken to monitor levels so as not to push them into estrogen dominance. The same situation is seen in menopause, as well.
Overall, remediation efforts directed at AFS with any kind of supplementation must be regarded with the entire set of body organ systems in mind. The most appropriate approach with which to do this is the NeuroEndoMetabolic (NEM) model of stress response.
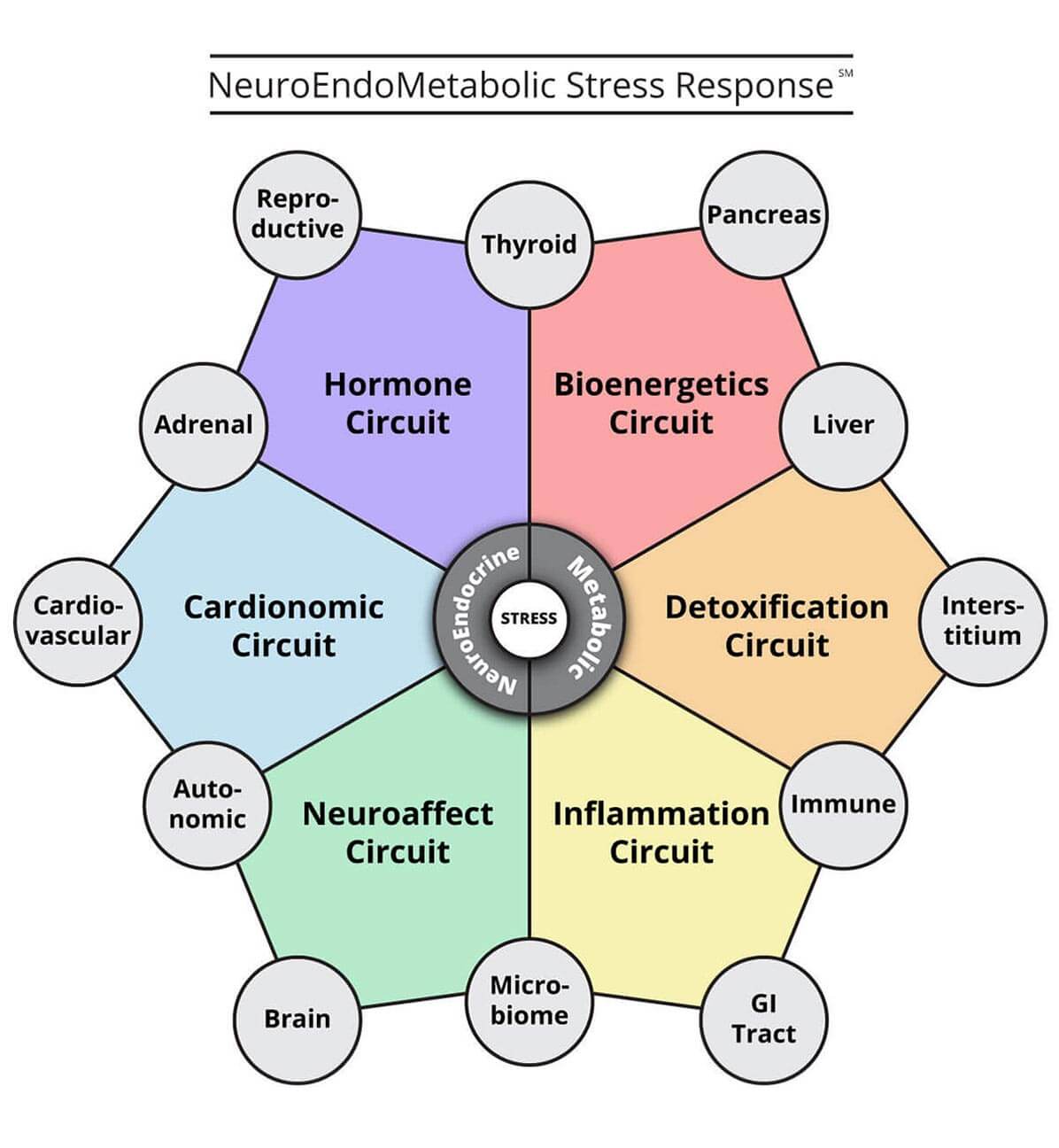
Traditionally trained physicians typically view symptoms as set apart from all but possibly one or two organs in the body. This is their approach in which only the symptoms are dealt with. The root causes of the condition for which their patient is seeking relief are not assessed.
On the other hand, practitioners who use the NEM model look at the six major organ systems and their inter-relationships in order to fully address the issues brought to them by their patients. This model says what affects one of these major organ systems affects others as well, including hypothalamus hormones systems.
In the case of women in menopause with AFS, the hormonal system will be affected greatly by fluctuating levels of estrogen, progesterone, and testosterone. The symptoms arising in this system will lead to fatigue, infertility, hair loss, low libido, and irregular menses.
With AFS, estrogen dominance can easily become a significant issue. Increasing stress puts more burden on the adrenals to secrete cortisol, which requires progesterone for production. This lowers the level of the hormone needed to balance estrogen. More inflammation also results from estrogen dominance.
Inflammation is also a product of metabolic system issues in AFS. With leaky gut syndrome, the toxins that enter the bloodstream cause more inflammation and an immune system response. This disruption in the metabolic system leads to dyslipidemia, central obesity, and the development of Type 2 diabetes.
With more women reaching the age of menopause and living quite a few years afterward, the issue of hormone replacement therapy grows in significance. The WHI results led to a cessation of synthetic HRT. More current research and clinical practice have shown HRT, both synthetic or bioidentical, to be effective on hypothalamus hormones and have its place when used correctly. Not only will this approach help women deal with the issues related to menopause, it also addresses issues related to AFS. Careful consideration of whether or not to use HRT should be entered into by women and their healthcare professionals.
Estrogen and other hypothalamus hormones have been labeled as dangerous by the researchers associated with the Women’s Health Initiative. Current research strongly refutes these results. This current research indicates estrogen replacement to be safe and very effective in alleviating menopausal symptoms and those of AFS.